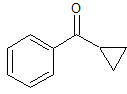

Ten g. of (3-chloropropyl)(phenyl)ketone was mixed with a solution of 11.3 g. of potassium hydroxide in 130 cc. of methyl alcohol; after 18 hours, about half the alcohol was distilled and the residue diluted with water and extracted with ether. The ether was dried and distilled and the residual oil distilled under diminished pressure, passing over almost completely at 140—142° (37 mm.); yield, 5.5 g. Its identity as benzoyl-cyclopropane was established by the following experiments.
a. On treatment with semicarbazide hydrochloride and potassium acetate in alcohol at room temperature for 12 hours, a solid product was obtained which after crystallization melted at 182—183° (Kishner gives 185°).
b. Treated with hydroxylamine hydrochloride and potassium hydroxide in alcohol at room temperature, an oil was obtained which soon crystallized and after recrystallizing from petroleum ether melted at 90—94° (Perkin gives 90—92°; Kishner, 86—88°).
c. Bromine in carbon tetrachloride was without action on the substance except in the sunlight when the bromine color slowly disappeared and hydrobromic acid was evolved.
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru