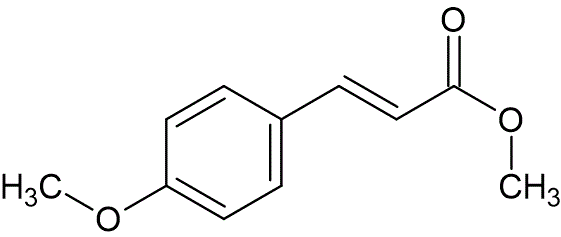

To a solution of methyl(triphenylphosphoranylidene)acetate (25 g, 75.7 mmol) in tetrahydrofuran (68 mL) at 25 °C, was added p-anisaldehyde (6.78 g, 49.8 mmol). The solution was heated at reflux for 4 hours, and then evaporated under reduced pressure to afford an impure residue. This material was purified by column chromatography, eluting with petroleum ether : ethyl acetate (90 : 10) to afford methyl 4-methoxycinnamate (9.0 g, 94%). This solid was recrystallized from methanol to provide pure ester, mp 87—89 °C, lit. mp 89 °C.
Rf 0.32 petroleum ether : ethyl acetate (90 : 10).
HRMS calcd. for C11H12O3 (M+H): 192.079. Found: 192.082.
IR (CHCl3): 3020 (weak), 2970 (weak), 2855 (weak), 1710 (strong), 1640 (medium), 1610 (medium), 1580 (medium), 1512 (medium), 1464 (weak), 1440 (medium), 1320 (medium), 1408 (medium), 1390 (medium), 1260 (strong), 1170 (strong), 1110 (weak), 1034 (medium), 985 (medium), 937 (weak), 860 (weak), 850 (weak), 830 (medium) cm-1.
1H NMR (CDCl3): δ 3.79 (3H, s), 3.83 (3H, s), 6.31 (1H, d, J = 15.9 Hz), 6.90 (2H, d, J = 8.8 Hz), 7.47 (2H, d, J = 8.8 Hz), 7.65 (1H, d, J = 15.9 Hz).
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru