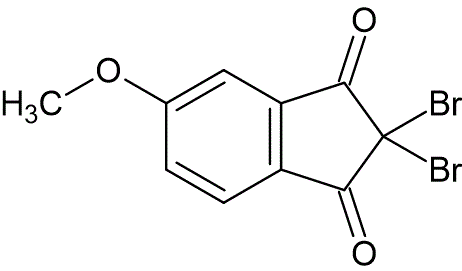

To a solution of 5-methoxy-1,3-indanedione (0.50 g, 2.8 mmol) in dioxane (15 mL) at 15 °C, was added pyridine (0.69 mL, 8.5 mmol), followed by bromine (0.32 mL, 6.2 mmol). This mixture was allowed to warm to 25 °C. After two hours the reaction was diluted with water (35 mL), and a yellow solid precipitated. This product was removed by filtration and dried under reduced pressure, to provide 2,2-dibromo-5-methoxy-1,3-indanedione (0.90 g, 95% yield). Recrystallization from methanol and water afforded pure product, mp 137—140 °C.
Rf0.36 petroleum ether : ethyl acetate (80 : 20).
IR (CHCl3): 3030 (weak), 2990 (weak), 2960 (weak), 2860 (weak), 1770 (medium), 1730 (strong), 1610 (strong), 1585 (strong), 1495 (medium), 1460 (weak), 1440 (weak), 1360 (weak), 1350 (weak), 1290 (strong), 1265 (strong), 1170 (weak), 1100 (medium), 1080 (medium), 1020 (medium), 990 (weak), 905 (weak), 890 (weak), 870 (medium) 850 (medium) 640 (medium) cm-1.
1H NMR (CDCl3): δ 4.05 (3H, s), 7.4 (1H, d, J = 2.3 Hz), 7.45 (1H, dd, J1 = 2.3 Hz, J2 = 8.1 Hz), 8.00 (1H, d, J = 8.1 Hz).
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru