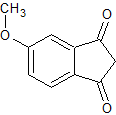

To a mixture of sodium hydride (60% oil dispersion, 2.14 g, 89.2 mmol) and dimethyl 4-methoxyphthalate (2.0 g, 8.9 mmol) in tetrahydrofuran (6 mL), at 25 °C, was added ethyl acetate (15.7 g, 0.178 mol). This mixture was heated to reflux. After 12 hours, the reaction was cooled to 25 °C, and then diluted with ethyl ether (20 mL) to form a slurry, from which a crude yellow-brown solid was obtained by filtration. The solid was dissolved in water (100 mL), acidified with 2N hydrochloric acid, and heated to 80 °C for 15 minutes. After cooling, a pale yellow solid precipitated. This was removed by filtration, and dried over P2O5 under reduced pressure, to afford 5-methoxy-1,3-indanedone (0.25 g, 16% yield). Recrystallization from methanol provided pure dione, mp 118—120 °C, lit. mp 118—119 °C.
Rf 0.77 methylene chloride : methanol (90 : 10).
IR (CHCl3): 3025 (weak), 2990 (weak), 2960 (weak), 2860 (weak), 1750 (strong), 1710 (strong), 1600 (strong), 1590 (strong), 1490 (medium), 1460 (medium), 1450 (strong), 1370 (weak), 1350 (medium), 1330 (weak), 1295 (strong), 1270 (strong), 1170 (weak), 1110 (weak), 1080 (medium), 1020 (medium), 880 (weak), 860 (medium), 840 (medium) cm-1.
1H NMR (CDCl3): δ 3.23 (2H, s), 3.96 (3H, s), 7.32-7.36 (2H, m), 7.87-7.91 (1H, m).
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru