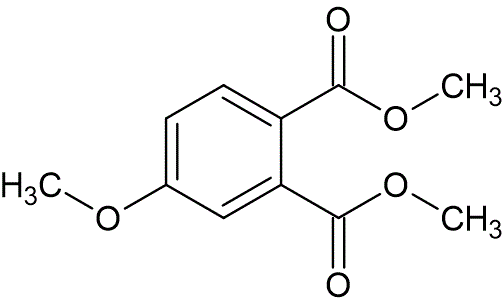

To a suspension of sodium hydride (60% oil dispersion, 0.65 g, 27.2 mmol) in tetrahydrofuran (10 mL), at 5 °C, was added dimethyl 4-hydroxyphthalate (3.81 g, 18.1 mmol), dissolved in a mixture of tetrahydrofuran (21 mL), and dimethyl formamide (3.5 mL). After addition of methyl iodide (5.14 g, 36.2 mmol) at 5 °C, the mixture was allowed to warm to 25 °C and was stirred for 12 hours. The reaction was cooled to 5 °C, and saturated ammonium chloride (60 mL) was added. The layers were separated, and the aqueous layer extracted with methylene chloride (30 mL). The organic phases were combined, dried over Na2SO4, filtered, and evaporated under reduced pressure to afford a crude oil. This oil was purified by column chromatography by elution with petroleum ether : ethyl acetate (80 : 20 and 70 : 30) to provide pure dimethyl 4-methoxyphthalate as an oil (3.17 g, 78% yield).
Rf 0.74 petroleum ether : ethyl acetate (50 : 50).
IR (CHCl3): 3020 (medium), 2980 (medium), 2855 (weak), 1720 (strong), 1610 (strong), 1580 (medium), 1500 (weak), 1460 (medium), 1450 (medium), 1440 (medium), 1330 (strong), 1300 (strong), 1240 (strong), 1030 (strong), 1075 (medium), 1035 (medium), 980 (weak), 965 (weak), 895 (weak), 880 (weak), 860 (weak), 840 (weak),830 (weak), 820 (weak) cm-1.
1H NMR (CDCl3): δ 3.86 (3H, s), 3.87 (3H, s), 3.92 (3H, s), 6.99 (1H, dd, J1 = 2.6 Hz, J2 = 8.6 Hz), 7.07 (1H, d, J = 2.6 Hz), 7.82 (1H, d, J = 8.6 Hz)
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru