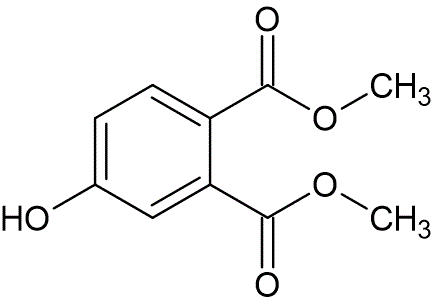

A solution of 1-methoxy-3-(trimethylsilyloxy)-1,3-butadiene (8.86 g, 51.4 mmol) and dimethyl acetylenedicarboxylate (4.87 g, 34.3 mmol) in benzene (25 mL) was heated to reflux. After six hours the reaction was concentrated under reduced pressure, and the crude residue was dissolved in tetrahydrofuran (35 mL), followed by the addition of 0.1N aqueous hydrochloric acid (35 mL). After 45 minutes, the reaction was made basic with saturated NaHCO3, and extracted with chloroform (100 mL, 3x). The organic layers were combined, dried over Na2SO4, filtered and concentrated under reduced pressure. The resulting crude oil was dissolved in ethyl ether, and upon addition of excess hexane, the diester precipitated and was collected by filtration to afford 5.3 g of a white solid (74% yield). This solid was recrystallized from a toluene : hexane mixture, to afford the pure product, mp 104.5—106.0 °C, lit. mp 107—108 °C.
Rf 0.56 petroleum ether : ethyl acetate (50 : 50).
IR (CHCl3): 3580 (weak), 3380 (weak, broad), 3020 (weak), 2960 (weak), 1720 (strong), 1610 (medium), 1580 (medium), 1435 (medium), 1295 (strong), 1125 (medium), 1070 (medium), 980 (weak), 960 (weak) cm-1.
1H NMR (CDCl3): δ 3.86 (3H, s), 3.90 (3H, s), 6.92 (1H, dd, J1 = 2.5 Hz, J2 = 8.5 Hz), 7.0 (1H, d, J = 2.5 Hz), 7.25-7.5 (1H, br, D2O exchangeable), 7.73 (1H, d, J = 8.5 Hz).
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru