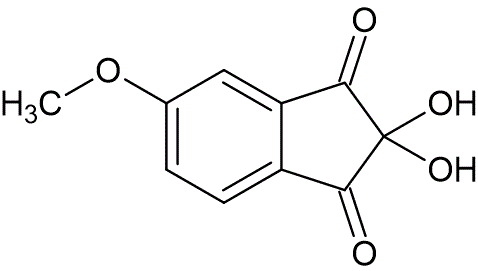

To a solution of 3-bromo-6-methoxy-1-indenone (4.78 g, 19.9 mmol) in benzene (95 mL) at 5 °C was added dimethylsulfoxide (95 mL), followed by the addition of bromine (1.59 g, 9.95 mmol). The solution was heated at reflux, and after six hours was cooled to 25 °C. The reaction was extracted with water (100 mL, 2x), and the aqueous washes were combined. The aqueous phase was then extracted with ethyl acetate (100 mL, 4x). The organic extracts were combined, washed with water (55 mL, 2x), dried over Na2SO4, filtered, and evaporated under reduced pressure to afford 5-methoxyninhydrin as an oil (2.90 g, 70% yield). The oil was dissolved in water by heating to reflux, then was allowed to crystallize overnight at 5 °C. Solid compound was obtained (2.43 g, 58% yield) as a white powder, mp 95—99 °C, lit. mp 80—85 °C (impure).
Rf 0.46 petroleum ether : ethyl acetate (50 : 50).
IR (CHCl3): 3500-2200 (broad), 1748 (strong), 1700 (strong), 1595 (strong), 1576 (medium), 1495 (medium), 1455 (weak), 1446 (weak), 1400 (medium), 1300 (strong), 1238 (medium), 1200 (weak), 1185 (medium), 1150 (medium). 1110 (medium), 1095 (medium), 1030 (medium), 1015 (medium), 975 (medium), 920 (medium), 880 (weak), 865 (medium), 800 (weak), 780 (medium), 683 (weak), 670 (weak) cm-1.
1H NMR (DMSO-d6): δ 3.98 (3H, s), 7.42 (1H, d, J = 2.4 Hz), 7.46 (2H, s, D2O exchangeable), 7.56 (1H, dd, J1 = 2.4 Hz, J2 = 8.5 Hz), 7.96 (1H, d, J = 8.5 Hz).
A solution of 5-methoxy-2,2-dibromo-1,3-indanedione (0.80 g, 2.4 mmol) in toluene (4.0 mL) was heated to 100 °C, followed by addition of dimethylsulfoxide (4.8 mL). After 3 hours, the reaction was cooled to 25 °C, diluted with water (10 mL), and extracted with petroleum ether (10 mL, 2x). The aqueous phase was then extracted with ethyl acetate (10 mL, 4x). The organic extracts were combined, washed with water (5 mL), dried over Na2SO4, filtered, and evaporated under reduced pressure to afford 5-methoxy-2,2-dihydroxy-1,3-indanedione as an oil (0.46 g, 95% yield). The oil was dissolved in water by heating at reflux, and was allowed to solidify overnight at 5 °C to afford 5-methoxyninhydrin (0.29 g, 59% yield) as a white powder. The spectral and physical data for this product were identical to those reported earlier.
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru