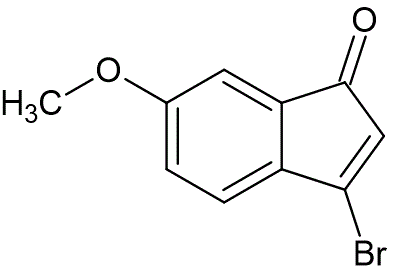

To a solution of 6-methoxy-1-indanone (0.50 g, 3.0 mmol) in carbon tetrachloride (20 mL), at 25 °C, was added N-bromosuccinimide (1.12 g, 63.0 mmol), and azobisisobutyronitrile (11 mg). The mixture was heated at reflux and illuminated with a 125W light. After one hour the reaction was cooled to 5 °C, and triethylamine (1.52 mL, 10.5 mmol) was added. The reaction mixture was allowed to stir for one hour at 25 °C. Solids were removed by filtration, and washed with carbon tetrachloride. The filtrate was washed with 2N HCl (50 mL), followed by water (50 mL, 3x), dried over Na2SO4, filtered, then evaporated under reduced pressure to afford pure 3-bromo-6-methoxy-1-indenone (0.72 g, 99%) as an orange solid, mp 100—102 °C.
Rf 0.49 petroleum ether : ethyl acetate (90 : 10).
HRMS calcd. for C10H7O2Br (M+): 237.961. Found: 237.965.
IR (CHCl3): 3420 (weak), 3140 (weak), 3030 (weak), 2990 (weak), 2965 (weak), 2860 (weak), 1710 (strong), 1600 (strong), 1533 (medium), 1477 (medium), 1435 (medium), 1360 (medium), 1345 (weak), 1285 (strong), 1253 (strong), 1160 (weak), 1100 (weak), 1047 (strong), 1022 (medium), 930 (weak), 804 (weak), 875 (weak), 822 (strong) cm-1.
1H NMR (CDCl3): δ 3.84 (3H, s), 6.09 (1H, s), 6.85 (1H, dd, J1 = 2.4 Hz, J2 = 8.0 Hz), 7.01 (1H, d, J = 2.4 Hz), 7.08 (1H, d, J = 8.0 Hz).
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru