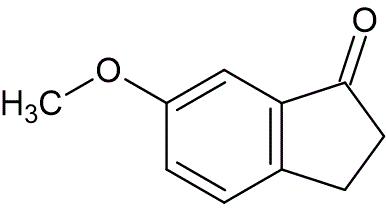

To a rapidly stirring solution of polyphosphoric acid (300 g) at 120 °C, was added 4-methoxydihydrocinnamic acid (3.1 g, 17.2 mmol). The reaction was allowed to proceed for five minutes, then quenched by pouring it on ice (1 Kg). The organic layer was separated and the aqueous layer extracted with methylene chloride (100 mL). The organic layers were combined, washed with 5% aqueous Na2CO3, dried over Na2SO4, and filtered. The filtrate was evaporated under reduced pressure to afford a crude residue, which was purified by column chromatography by eluting with petroleum ether : ethyl acetate (90 : 10). Pure 6-methoxy-1-indanone (1.75 g, 63%) was obtained as a white solid, mp 107—109 °C, lit. mp 108—109 °C.
Rf0.41 petroleum ether : ethyl acetate (80 : 20).
IR (CHCl3): 3010 (medium), 2973 (medium), 2968 (medium), 2880 (weak), 2850 (weak), 1705 (strong), 1616 (medium), 1588 (weak), 1493 (strong), 1468 (medium), 1444 (medium), 1435 (medium), 1405 (weak), 1338 (medium), 1320 (weak), 1300 (strong), 1280 (strong), 1250 (medium), 1190 (weak), 1165 (medium), 1145 (strong), 1128 (medium), 980 (weak), 894 (weak), 862 (weak), 836 (medium) cm-1.
1H NMR (CDCl3): δ 2.69-2.74 (2H, m), 3.05-3.09 (2H, m), 3.84 (3H, s), 7.16-7.20 (2H, m), 7.35-7.39 (1H, m).
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru