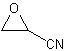
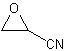
A 32.8-g. (0.25 mole) sample of glycidaldehyde oxime acetate was charged to a 100-ml distillation kettle and attached to a 0.7 X 50 cm glass spiral-packed column. The pressure was held at 50 mm while heating with an oil-bath. Deacetylation started when the bath temperature reached 115-120 C, but in order to obtain a satisfactory distillation rate it was necessary to hold the bath at 140-145 C. Takeoff was adjusted to maintain a maximum head temperature of 70 C. After 2.5 hr, 24.8 g of a mixture of acetic acid and glycidonitrile had been collected and the rate of takeoff was very slow; by lowering the pressure to 1 mm only 0.4 g more material distilled as a separate fraction. The distillation residue was 5.0 g.
Careful distillation of the mixture of acid and nitrile through the same column afforded 7.3 g. (42% yield) of glycidonitrile, b.p. 47.5-48 C (20 mm.), n (20 C, D) 1.4094. Infrared analysis showed nitrile absorption at 4.43 um and epoxide absorption at 8.05 and 11.98 um.
The epoxy nitrile was also isolated by dissolving 69 g of the mixture of acetic acid and glycidonitrile in 300 ml of ether and washing out the acetic acid with a concentrated sodium bicarbonate solution. Distillation of the dried ether solution gave 21 g of glycidonitrile with the above b.p.
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru